Welcome to Akshar Shop
Sodium Nitrate
Sodium Nitrate
Sodium nitrate should not be confused with sodium nitrite, which is also a common food additive and preservative used, for example, in deli meats. Less common applications include as an oxidizer in fireworks, replacing potassium nitrate commonly found in black powder, and as a component in instant cold packs.
What is Sodium Nitrate?
Sodium nitrate is an inorganic nitrate salt of an alkali metal with the chemical formula NaNO3.
Commonly referred to as “Chile saltpeter”, this compound consists of a sodium cation (Na+) and a nitrate anion (NO3–). At room temperature, sodium nitrate exists as a white, crystalline solid which is highly soluble in water
This compound is non-flammable. However, it is a strong oxidizing agent and can react with many flammable compounds in a violent manner. NaNO3 decomposes explosively when heated to temperatures above 538oC.
Rich deposits of sodium nitrate can be found in some South American countries such as Chile and Peru. The primary applications of this compound are in agriculture (fertilizers) and pyrotechnics.
Sodium Nitrate Structure
Sodium nitrate features an ionic bond between one Na+ ion and one NO3– ion. The structure of a NaNO3 molecule is illustrated below.
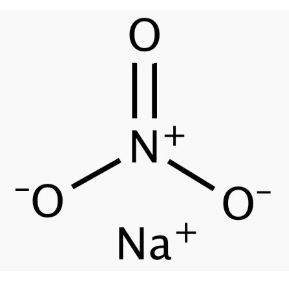
The nitrate anion has a trigonal planar structure in which 3 oxygen atoms are bonded to a central nitrogen atom. The negative charge on this ion is delocalized due to resonance. Therefore, the nitrogen atom a charge of +1 whereas each oxygen atom carries a charge of -⅔. The net formal charge on the NO3– is -1
Physical Properties
- Sodium nitrate is a crystalline solid which is white in color.
- It has two crystal structures – rhombohedral and trigonal.
- This compound has a sweet odor.
- The solubility of NaNO3 in water corresponds to 91.2g/100mL at a temperate of 25o
- This compound is also highly soluble in ammonia
Feature:
Uses of Sodium Nitrate
Owing to its high solubility in water, low cost, and nitrogen content, sodium nitrate is used in several fertilizers. Some other uses of this compound are listed below.
- Hybrid forms of aqua regia can be prepared with the help of NaNO3. These hybrids also have the ability to dissolve gold.
- This compound is widely used as a food additive since it acts as a preservative.
- Sodium nitrate is used as an oxidizer in several types of fireworks.
- It is also a component of some instant cold packs.
- NaNO3 is one of the components used for the storage and transfer of heat in some solar power plants.
- In order to promote the growth of Nitrosomonas bacteria, this compound is added to the wastewater in several wastewater treatment plants.
Sodium nitrate is also used in several rocket propellants and is known to be a substitute for potassium nitrate in gunpowder.








