Welcome to Akshar Shop
Sodium Phosphate
Sodium Phosphate
SHMP is used as a sequestrant and has applicationswithin a wide variety of industries, including as a food additive in which it is used under the E number E452i.Sodium carbonate is sometimes added to SHMP to raise the pH to 8.0–8.6, which produces a number ofSHMP products used for water softening and detergents.
What is Sodium Phosphate?
Sodium phosphate is also known as phospho soda with the formula Na3PO4 is a saline cathartic that is familiar to radiologists since it is often used as a cleansing agent prior to double contrast barium enema.
Sodium phosphate cradles are the most widely recognized, yet there is broad utilization of potassium phosphate buffer and blends of sodium and potassium. Many compounds of pharmaceutical interest are formulated in sodium phosphate buffers.
Sodium Phosphate Structure – Na3PO4

Feature:
Uses of Sodium Phosphate – Na3PO4
- Short-term, local treatment of inflammation with neomycin as bacterial prophylaxis.
- Used after glaucoma surgery or after cataract surgery.
- Used as a mild laxative, stimulates emptying of gall-bladder.
- One of the most palatable of the saline laxatives. It is also used in the form of the oral solution (see below) as an antihypercal- cemic.
- Used to control the pH of water hardness precipitation and control agent in mildly acidic solutions.
Frequently Asked Questions
Is sodium phosphate an acid or a base?
Disodium hydrogen phosphate is a chemical compound with the formula Na2HPO4, also known as disodium phosphate. It’s more neutral (not acidic, or fundamental). This is used to prevent food from clumping. It is made with phosphorus by reaction of some sodium hydroxide.
What is sodium phosphate used for in medicine?
Sodium biphosphate and sodium phosphate are sources of phosphorus which is a material that occurs naturally and is essential in every cell in the body. Sodium biphosphate and sodium phosphate is a combination drug used in adults before a colonoscopy to relieve constipation and cleanse the intestines.
What happens if your phosphate levels are low?
High phosphate levels seldom contribute to hypophosphateemia symptoms; rather symptoms generally arise from the underlying disorder that causes hypophosphateemia. Ultra low levels of phosphate can cause difficulty breathing, agitation, altered mental state, muscle weakness and muscle damage called rhabdomyolysis.
What is sodium phosphate monobasic monohydrate?
Sodium Phosphate, Monobasic (monohydrate) is a reagent commonly used in molecular biology, biochemistry, and chromatography with very high buffering capacity. Monobasic sodium phosphate is extremely hygroscopic, and soluble in water.
What is the difference between sodium phosphate monobasic and dibasic?
Sodium phosphate monobasic has the chemical formula of NaH2PO4, and the chemical formula of Na2HPO4 has the sodium phosphate dibasic. As sodium phosphate dibasic dissolves in water the basicity in the medium is higher than when monobasic sodium phosphate dissolves in water.
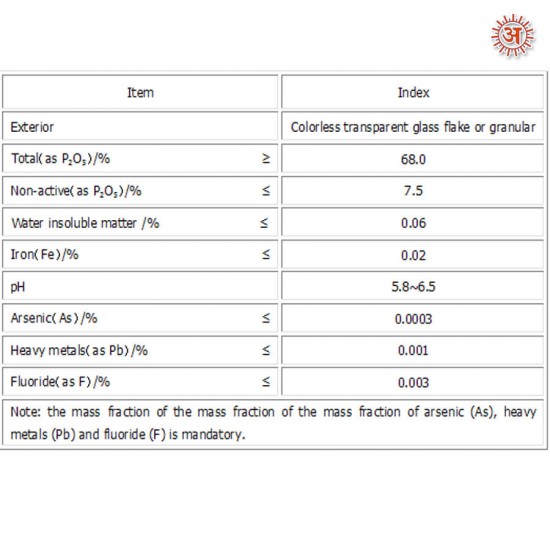
Specification
- Na3PO4 : Sodium Phosphate
- Density : 1.62 g/cm³
- Molecular Weight/ Molar Mass : 163.94 g/mol
- Boiling Point : 100 °C
- Melting Point : 1,583 °C
- Chemical Formula : Na3PO4
- Odour : Odorless
- Appearance : Off-white powder
- Covalently-Bonded Unit : 4
- Complexity : 36.8
- Hydrogen Bond Acceptor : 4
- Solubility : Soluble in water